|
This fact has key implications for the building up of the periodic table of elements. The ordering of the electrons in the ground state of multielectron atoms, starts with the lowest energy state (ground state) and moves progressively from there up the energy scale until each of the atom’s electrons has been assigned a unique set of quantum numbers. 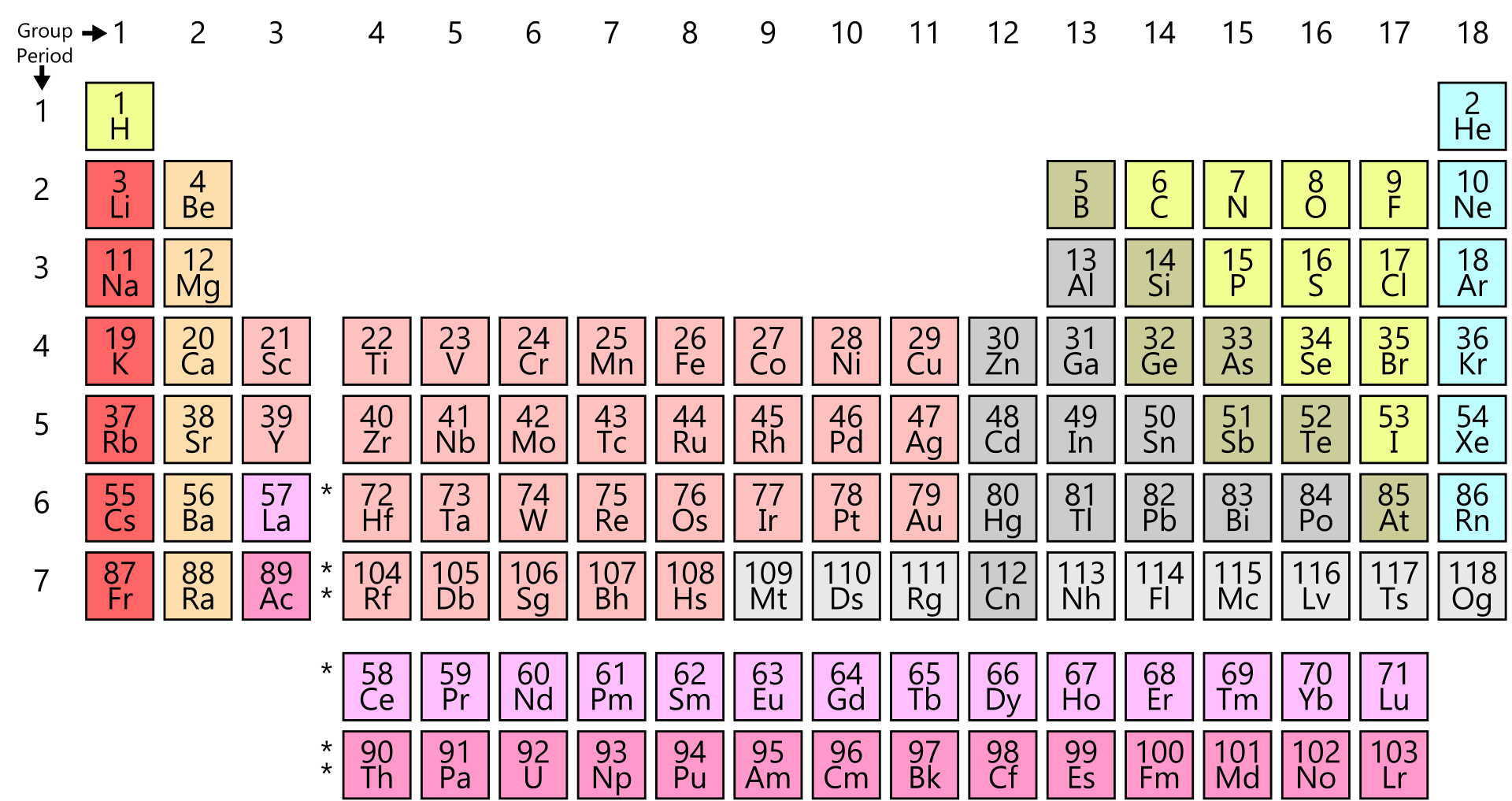
Transuranic elements have atomic number > 92. It is the Pauli exclusion principle that requires the electrons in an atom to occupy different energy levels instead of them all condensing in the ground state. Lessons on the periodic table for high school chemistry experiments. In the periodic table, the elements are listed in order of increasing atomic number Z. The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. The configuration of these electrons follows from the principles of quantum mechanics. The chemical properties of the atom are determined by the number of protons, in fact, by number and arrangement of electrons. See also: Atomic Number – Does it conserve in a nuclear reaction? Atomic Number and Chemical PropertiesĮvery solid, liquid, gas, and plasma is composed of neutral or ionized atoms. It is the electrons that are responsible for the chemical bavavior of atoms, and which identify the various chemical elements. In a neutral atom there are as many electrons as protons moving about nucleus. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The nucleus is composed of protons and neutrons. Charts come from empirical data on the real behavior of elements, which may differ somewhere from the periodic table predictions. When an electron loses energy (and so emits a photon), it might migrate to an inner shell that isn't completely occupied.The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Group 17 (Halogens): 1- Group 18 (Noble Gases): 0 Use a chart. Like many others of my generation, I too had crammed this up during my high school. Chlorine, symbol Cl, has a Base Centered Orthorhombic structure and. 
Alternatively, the electron can break free from its linked atom's shell, resulting in ionisation and the formation of a positive ion. Btw - this is how a Periodic Table looks like. Element 17 of Periodic table is Chlorine with atomic number 17, atomic weight 35.453. Atomic excitation occurs when an energy gain causes an electron to migrate (jump) to an outer shell. There are eight valence electrons in all.Ī valence electron may receive or release energy in the form of a photon, much like a core electron. Here is the electrical configuration of chlorine. You can find a periodic table online or in a chemistry book. Other elemental information includes atomic weight and atomic number. Element number 17 of the Periodic Table of the Elements - Chemistry - Magenta square frame with black background Stock Vector and. It is color-coded and assigns each element a unique 1 or 2-letter abbreviation. Valence electrons are electrons in the outer shells that are not completely filled. Many periodic tables list numbers for element groups, which are columns of the periodic table. The periodic table is a chart that organizes elements by their atomic structure. In this sense, the reactivity of a particular element is strongly reliant on its electrical arrangement. The existence of valence electrons can affect an element's chemical characteristics, such as its valence-whether or not it can bind with other elements and, if so, how easily and how many times. 
At room temperature, chlorine is a pale green gas with a yellow-green colour.Ī valence electron is an outer shell electron connected with an atom that can participate in the creation of a chemical bond if the outer shell is not closed in a single covalent connection, both atoms contribute one valence electron to create a shared pair in chemistry and physics. It is the second-lightest of the halogens, appearing on the periodic table between fluorine and bromine, and its characteristics are largely midway between them. Atomic number: 17: Atomic mass amu 35.453: Atomic mass pm 102: Density at STP g/cm3 0.0032: Number of protons: 17: Number of neutrons (typical isotopes) 35 37: Number of electrons: 17: Electron configuration. 
Hint: Chlorine is a chemical element with the atomic number 17 and the symbol Cl.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
